Publications of 2026
“Fast, selective and scalable flow ammonolysis of oxiranes accessible from glycerol toward bio-based amines“
H. L. Lee, F. Molajafari, A. Koishybay, Y. Miyai, L. Zimmermann, J.-C. M. Monbaliu, C. Vlaar, R. J. Romañach, Md. Noor-E-Alam, A. S. Myerson, T. Stelzer
Org. Proc. Res. Dev. 2026, Accepted
“Fast, selective and scalable flow ammonolysis of oxiranes accessible from glycerol toward bio-based amines“
F. Barbaz, H. Hellwig, D. V. Silva-Brenes, J.-C. M. Monbaliu
Green Chem. 2026, 28, 4982-4989
“Controlling Gold Nanoparticles Aggregation in Continuous-Flow Microreactors for Robust Production of Ultrasensitive NIR SERS Nanotags“
A. Verdin, P. Stiernet, L. Lejeune, J.-C. M. Monbaliu, S. Hermans, G. Eppe and C. Malherbe
ACS Applied Nano Materials 2026, 2, 1307-1318
“Nemo Censetur Ignorare Legem: What Research Chemists Should Know About the EU Legal Framework“
S. Bianchi,= C. Campalani,= V. Vella, Q. Michel, and J.-C. M. Monbaliu*
Chem. Soc. Rev. 2026, 55, 1813-1861
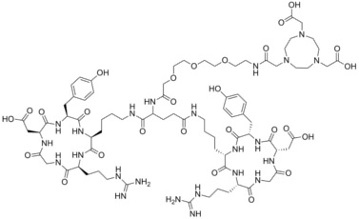
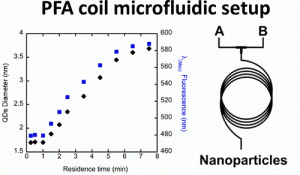
“Aqueous Continuous Flow Synthesis of Cadmium Chalcogenide Quantum Dots: Opportunities and Challenges”
C. Campalani, G. Petit, and J.-C. M. Monbaliu*
JACS Au 2026, 1, 38-58
Publications of 2025
“A Multidimensional In Silico Guidance to Map and Optimize Competing Nitroso Reactions“
P. Bianchi, F. Mazars, S. Renson, and J.-C. M. Monbaliu*
ChemRxiv 2025
DOI: 10.26434/chemrxiv-2025-72s12
“Catalytic Generation of Nitrosocarbonyls under Mild Conditions: Advances, Challenges, and Opportunities“
Y.-H. Tsai, C. Campalani, T. Toupy, I. Jacquemin, and J.-C. M. Monbaliu*
ChemSusChem 2025, Accepted
“Controlled API crystallization during additive manufacturing of solid dosage form for flexible integrated pharmaceutical manufacturing“
A. Mbodji, K. Mulero Cruz, A. Arroyo Gómez, C. P. Vlaar, J. Duconge, J.-C. M. Monbaliu, R. K. Cersonsky, L. Yu, G. G.-Z. Zhang, G. Coquerel, R. J. Romanach, and T. Stelzer
Int. J. Pharm. 2025, 685, 126197
“Intensified Continuous Flow Synthesis of Oxazolidones“
L. Crane, T. Habets, B.Grignard, J.-C. M. Monbaliu*, P. Stiernet and C. Detrembleur
ACS Sustainable Chem. Eng. 2025, 13, 16107-16116
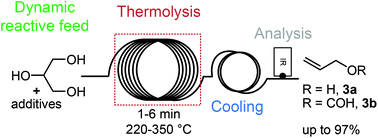
“Continuous Flow Synthesis of Nitrofuran Pharmaceuticals using Acetyl Nitrate”
H. Hellwig, L. Bovy, K. Van Hecke, C. P. Vlaar, R. J. Romañach, Md. Noor-E-Alam, A. S. Myerson, T. Stelzer, and J.-C. M. Monbaliu*
Angew. Chem. Int. Ed. 2025, 64, e202501660
2025 Flow Chemistry Hot Topic
“Open-source fraction collector for flash column chromatography and continuous flow reactions”
Y. Chen, C. Desmons, M. Cattoen, J.-C. M. Monbaliu*
React. Chem. Eng. 2025, 10, 1408-1416
“Photooxygenation Reactions of Olefins Under Flow Conditions: An Experimental and In-Silico Study”
G. Moroni, Y.-H. Tsai, M. Ballarotto, A. Carotti, J.-C. M. Monbaliu*, A. Gioiello*
ChemPhotoChem 2025, 9, 202500027
“Towards sustainable quantum dots: regulatory framework, toxicity and emerging strategies”
C. Campalani and J.-C. M. Monbaliu*
Mat. Sci. Eng. R. 2025, 163, 100940
“Oxidative neutralisation of sulfur-based chemical warfare agents mediated by a lipase: from batch to flow reactor”
M. Boddaert, V. Baptista da Silva, S. Mansour, D. Vuluga, P.-Y. Renard, J.-C. M. Monbaliu* and J. Legros*
Chem. Eur. J. 2025, 31, e202403701
“A review of precision medicine in developing pharmaceutical products: Perspectives and opportunities”
J. E. Martínez-Jiménez, I. Sathisaran, F. Reyes Figueroa, S. Reyes, M. López-Nieves, C. P. Vlaar, J.-C. M. Monbaliu, R. Romañach, G. Ruaño, T. Stelzer, and J. Duconge
Int. J. Pharm. 2025, 670, 125070
“Rapid CO2 coupling to propargylic alcohols: unlocking the production of α-alkylidene cyclic carbonates via continuous flow”
P. Stiernet, A. Verdin, M. Stina Svanberg Frisinger, B. Grignard, C. Malherbe, J. Yuan, J.-C. M. Monbaliu* and Christophe Detrembleur*
Green Chem. 2025, 27, 722-730
Publications of 2024
“Innovative Chalcogenide Transfer Agent for Improved Aqueous Quantum Dot Synthesis”
G. Petit, C. Malherbe, P. Bianchi and J.-C. M. Monbaliu*
Chem. Sci. 2024, 15, 13115-13560
Featured in the “Celebrating the scientific accomplishments of RSC Fellows” themed collection
Front cover: September 2024
“The Catalytic Coupling of CO2 and Glycidol Toward Glycerol Carbonate”
C. Muzyka, D. V. Silva-Brenes, B. Grignard, C. Detrembleur and J.-C. M. Monbaliu*
ACS Catalysis 2024, 14, 12454-12493
“Continuous Flow Synthesis and Crystallization of Modafinil: A Novel Approach for Integrated Manufacturing”
D. V. Silva-Brenes, S. Agrawal, V. López Mejías, J. Duconge, C. P. Vlaar, J.-C. M. Monbaliu* and Torsten Stelzer
React. Chem. Eng. 2024, 9, 2728-2739
“New Opportunities for Organic Synthesis with Superheated Flow Chemistry”
P. Bianchi and J.-C. M. Monbaliu*
Acc. Chem. Res. 2024, 57, 2207-2218
DOI: 10.1021/acs.accounts.4c00340
Front cover: August 2024
“Glycerol and its derivatives as potential C-3 bio-based building blocks for accessing active pharmaceutical ingredients”
L. Bovy, R. Morodo, D. V. Silva-Brenes and J.-C. M. Monbaliu*
Green Chem. 2024, 26, 10029-10057
Front cover: September 2024
“Continuous flow synthesis of functional isocyanate-free poly(oxazolidone)s by step-growth polymerization”
F. Siragusa, L. Crane, P. Stiernet, T. Habets, B. Grignard, J.-C. M. Monbaliu* and C. Detrembleur*
ACS Macro Lett. 2024, 13, 644-650
“On a seamlessly replicable circular photoreactor for lab-scale continuous flow applications”
Y.-H. Tsai, M. Cattoen, G. Masson, G. Christen, L. Traber, M. Donnard, F. Leroux, G. Bentzinger, S. Guizzetti and J.-C. M. Monbaliu*
React. Chem. Eng. 2024, 9, 1946-1655
Featured in the Special Edition celebrating Prof. K. F. Jensen’s 70th Birthday
Front cover: July 2024
“Preparation of Dinitrogen Trioxide for Organic Synthesis: A Phase Equilibrium Approach”
Y. Chen and J.-C. M. Monbaliu*
Chem. Eur. J. 2024, 30, e202303516
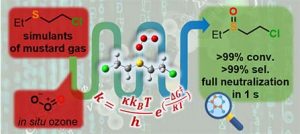
“A miniaturized ozonolysis flow platform for expeditious sulfur mustard warfare simulant neutralization”
M. Boddaert, P. Bianchi, D. V. Silva-Brenes, A. Musina, M. Winter, P. M. C. Roth, P.-Y. Renard, J. Legros and J.-C. M. Monbaliu*
Green Chem. 2024, 26, 1281-1288
Front cover: February 2024
Featured in the RSC Green Chemistry 25th anniversary themed collection
“Intensified continuous flow process for the scalable production of bio-based glycerol carbonate”
C. Muzyka, S. Renson, B. Grignard, C. Detrembleur, J.-C. M. Monbaliu*
Angew. Chem. Int. Ed. 2024, 63, e202319060
HOT Article 2024
“New insights into the role of VKORC1 polymorphisms for optimal warfarin dose selection in Caribbean Hispanic patients through an external validation of a population PK/P D model”
K. Rodríguez -Fernández, G. Reynaldo-Fernández, S. Reyes-González, C. de las Barreras, L. Rodríguez-Vera, C. Vlaar, J.-C. M. Monbaliu, T. Stelzer, J. Duconge, V. Mangas-Sanjuan
Biomed. Pharmacother. 2024, 170, 115977
Publications of 2023
“Intensified continuous flow process for the scalable production of bio-based glycerol carbonate”
C. Muzyka, S. Renson, B. Grignard, C. Detrembleur, and J.-C. M. Monbaliu*
ChemRxiv 2023
“Continuous flow intensification for the synthesis of high purity warfarin”
D. V. Silva-Brenes, S. K. Reyes-Vargas, J. Duconge, C. Vlaar, T. Stelzer, and J.-C. M. Monbaliu*
Org. Process. Res. Dev. 2024, 28, 1704-1712
“Revisiting the Paradigm of Reaction Optimization in Flow with a Priori Computational Reaction Intelligence”
P. Bianchi and J.-C. M. Monbaliu*
Angew. Chem. Int. Ed. 2023, 63, e202311526
Open access – free to read
Part of the themed collection: Hot Topic : Flow Chemistry
Part of the themed collection: Hot Topic : Artificial Intelligence and Machine Learning
Monthly Highlights from the Literature: Some Items of Interest to Process R&D Chemists and Engineers
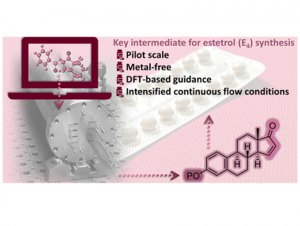
“Metal-free synthesis of estetrol key intermediate under intensified continuous flow conditions”
P. Bianchi, A. Dubart, M. Moors, D. Cornut, G. Duhirwe, J. Ampurdanés Vilanova, and J.-C. M. Monbaliu*
React. Chem. Eng. 2023, 8, 1565-1575
Front cover: June 2023
“A perspective on automated advanced continuous flow manufacturing units for the upgrading of bio based chemicals towards pharmaceuticals”
G. Kaisin*, L. Bovy, Y. Joyard, N. Maindron, V. Tadino, J.-C. M. Monbaliu*
J. Flow Chem. 2023, 23, 1349-1357
“Continuous Flow Photooxidative Degradation of Azo Dyes with Biomass-derived Carbon Dots”
C. Campalani, G. Petit, J.-C. M. Monbaliu*, M. Selva and A. Perosa*
ChemPhotoChem 2023, 7, e202200234
“Will the next generation of chemical plants be in miniaturized flow reactors?”
J.-C. M. Monbaliu* and J. Legros*
Lab Chip 2023, 23, 1349-1357
Collaborative Front cover: March 2023
Publications of 2022
“On Demand Flow Platform for the Generation of Anhydrous Dinitrogen Trioxide and Its Further Use in N-Nitrosative Reactions“
Y. Chen, S. Renson, J.-C. M. Monbaliu*
Angew. Chem. Int. Ed. 2022, 61, e202210146
Open access – free to read
Part of the themed collection: Hot Topic : Flow Chemistry
“Accelerating the End-to-end Production of Cyclic Phosphate Monomers with Modular Flow Chemistry“
R. Morodo, R. Riva, N. M. S. van den Akker, D. G. Molin, C. Jerome and J.-C. M. Monbaliu*
Chem. Sci. 2022, 13, 10699-10706
Open access – free to read
“Development and validation of an integrated microfluidic device with an in-line Surface Enhanced Raman Spectroscopy (SERS) detection of glyphosate in drinking water“
G. Emonds-Alt, C. Malherbe, A. Kasemiire, H. T. Avohou, P. Hubert, E. Ziemons, J.-C. M. Monbaliu, G. Eppe
Talanta 2022, 249, 123640
“A Continuous Flow Generator of Organic Hypochlorites for the Neutralization of Chemical Warfare Agent Simulants”
V.-E. H. Kassin, D. V. Silva-Brenes, T. Bernard, J. Legros, J.-C M. Monbaliu*
Green Chem. 2022, 24, 3167-3179
“Continuous flow organocatalyzed methoxycarbonylation of benzyl alcohol derivatives with dimethyl carbonate”
T. Toupy, L. Bovy, J.-C M. Monbaliu*
J. Flow Chem. 2022, 12, 207–217
“Out-smarting smart drug modafinil through flow chemistry”
D. V. Silva-Brenes, N. Emmanuel, V. Lopez-Mejias, J. Duconge Soler, C. Vlaar, T. Stelzer, J.-C M. Monbaliu*
Green Chem. 2022, 24, 2094–2103
“Intensified Continuous Flow Michaelis–Arbuzov Rearrangement toward Alkyl Phosphonates”
T. Toupy, J.-C M. Monbaliu*
Org. Process Res. Dev. 2022, 26, 467–478
“Perspectives for the Upgrading of Bio-based Vicinal Diols within the Developing European Bioeconomy”
C. Muzyka, J.-C M. Monbaliu*
ChemSusChem 2022, 15, e202102391
Cover feature: May 2022
“A multifaceted approach towards understanding the peculiar behavior of (?)-hydroxyiminophosphonates”
T. Toupy, C. Kune, K. Van Hecke, L. Quinton,* J.-C M. Monbaliu*
Org. Chem. Front. 2022, 9, 173-182
“Three decades of unveiling the complex chemistry of C-nitroso species with computational chemistry”
P. Bianchi, J.-C. M. Monbaliu
Org. Chem. Front. 2022, 9, 223-264
Front cover: January 2022 issue 1
Publications of 2021
“Identification of the degradation products from alpha-ionone used as stabiliser in “green” propellants through its lifetime”
C. Damseaux,* G. Scholl, R. Dobson, A. Dejeaifve, X. Ma, I. E. Marko, C. Damblon, J.-C. M. Monbaliu, E. De Pauw, G. Eppe
Propellants Explos. Pyrotech. 2021, 46, 1653-1661
“Flow neutralization of sulfur-containing chemical warfare agents with Oxone: packed-bed vs. aqueous solution”
A. Delaune, S. Mansour, B. Picard, P. Carrasqueira, I. Chataigner, L. Jean, P.-Y. Renard, J.-C. M. Monbaliu and Julien Legros*
Green Chem. 2021, 23, 2925-2930
“A Modular, Low Footprint and Scalable Flow Platform for the Expedient alpha-Aminohydroxylation of Enolizable Ketones”
V.-E. Kassin, R. Morodo, T. Toupy, I. Jacquemin, K. Van Hecke, R. Robiette and J.-C. M. Monbaliu*
Green Chem. 2021, 23, 2336-2351
Inside Front cover: March 2021 issue 21
Highlighted in: OPRD (Some items of Interest to Process R&D Chemists and Engineers)
Publications of 2020
“Au Nanobipyramids@mSiO2 Core-Shell Nanoparticles for Plasmon-Enhanced Singlet Oxygen Photooxygenations in Segmented Flow Microreactors”
C. Mendoza, A. Désert, D. Chateau, C. Monnereau, L. Khrouz, F. Lerouge, C. Andraud, J.-C. M. Monbaliu, S. Parola and B. Heinrichs
Nanoscale Adv. 2020, 2, 5280-5287
“Continuous Flow Upgrading of Selected C2-C6 Platform Chemicals Derived from Biomass”
R. Gérardy, D. P. Debecker, J. Estager, P. Luis and J.-C. M. Monbaliu*
Chem. Rev. 2020, 120, 15, 7219-7347
DOI: 10.1021/acs.chemrev.9b00846
“Continuous flow organophosphorus chemistry”
R. Morodo, P. Bianchi and J.-C. M. Monbaliu*
Eur. J. Org. Chem. 2020, 33, 5236-5277
Front cover : September 2020 issue 33
Tagged as Very Important Paper
Featured in the special Collection YourJOC Talents 2020
“A safe and compact flow platform for the neutralization of a mustard gas simulant with air and light”
N. Emmanuel, P. Bianchi, J. Legros and J.-C. M. Monbaliu*
Green Chem. 2020, 22, 4105-4115
Outside back cover: July 2020 issue 13
“Scalable and robust photochemical flow process towards small spherical gold nanoparticles”
P. Bianchi, G. Petit and J.-C. M. Monbaliu*
React. Chem. Eng. 2020, 5, 1224-1236
“The deoxydehydration (DODH) reaction: a versatile technology for accessing olefins from bio-based polyols”
N. Ntumba Tshibalonza and J.-C. M. Monbaliu*
Green Chem. 2020, 22, 4801-4848
Outside front cover: August 2020 issue 15
“Separation of bio-based chemicals using pervaporation”
W. Li, J. Estager, J.-C. M. Monbaliu, D. Debecker and P. Luis
J. Chem. Technol. Biotechnol. 2020, 95, 2311–2334
“Radiosynthesis of [18F]difluoromethyl heteroaryl-sulfones for radical C-H 18F-difluoromethylation of heteroarenes by visible light photoredox catalysis”
A Lemos, L. Trump, B. Lallemand, P. Pasau, J. Mercier, C. Lemaire, J.-C. M. Monbaliu, C. Genicot and A. Luxen
Catalysts 2020, 10, 275
“Metal-free hydroxylation of tertiary ketones under intensified and scalable continuous flow conditions”
V.-E. H. Kassin, T. Toupy, G. Petit, P. Bianchi, E. Salvadeo and J.-C. M. Monbaliu*
J. Flow Chem. 2020, 10, 167-179
“Supported ionic liquid membranes for the separation of methanol/dimethyl carbonate mixtures by pervaporation”
W. Li, C. Molina Fernandez, J. Estager, J.-C. M. Monbaliu, D. Debecker and P. Luis
J. Membr. Sci. 2020, 598, 117790
“Development of a sustainable continuous flow approach toward allantoin”
E. Salvadeo and J.-C. M. Monbaliu
J. Flow Chem. 2020, 10, 251-257
Publications of 2019
“Versatile and scalable synthesis of cyclic organic carbonates under organocatalytic continuous flow conditions”
R. Gérardy, J. Estager, P. Luis, D. P. Debecker and J.-C. M. Monbaliu*
Catal. Sci. Technol. 2019, 9, 6841-6851
Outside Front Cover: December 2019 issue 24
“Understanding chemical interaction between phosphonate-derivative molecules and silver surface cluster in SERS: a combined experimental and computational approach”
G. Emonds-Alt, B. Mignolet, C. Malherbe, J.-C. M. Monbaliu, F. Remacle and G. Eppe*
Phys. Chem. Chem. Phys. 2019, 21, 22180-22187
“Continuous flow upgrading of glycerol toward oxiranes and active pharmaceutical ingredients thereof”
R. Morodo, R. Gérardy, G. Petit and J.-C. M. Monbaliu*
Green Chem. 2019, 21, 4422-4433
“Expedient Preparation of Active Pharmaceutical Ingredient Ketamine under Sustainable Continuous Flow Conditions”
V.-E. Kassin, R. Gérardy, T. Toupy, D. Collin, E. Salvadeo, F. Toussaint, K. Van Hecke and J.-C. M. Monbaliu*
Green Chem. 2019, 21, 2952-2966
Part of the themed collection: 2019 Green Chemistry Hot Articles
Inside Front Cover: June 2019 issue 11
“Native chemical ligation and extended methodologies. Mecanisms, catalysis, scope and limitations”
V. Agouridas, O. El Mahdi, V. Diemer, M. Cargoët, J.-C. M. Monbaliu* and O. Melnyk*
Chem. Rev. 2019, 112, 7328-7443
“Solubility Determination and Correlation of Warfarin Sodium 2-Propanol Solvate in Pure, Binary, and Ternary Solvent Mixture”
M. V. George De la Rosa, R. Santiago, J. Malavé Romero, J. Duconge, J.-C. M. Monbaliu, V. López-Mejías, T. Stelzer
J. Chem. Eng. Data 2019, 64, 1399–1413
“Finding the perfect match: a combined computational and experimental study towards efficient and scalable photosensitized [2+2]cycloadditions in flow”
J. Williams, M. Nakano, R. Gérardy, J. A. Rincon, O. de Frutos, C. Mateos, J.-C. M. Monbaliu and C. O. Kappe
Org. Process Res. Dev. 2019, 23, 78-87
“Solvent-free organocatalytic preparation of cyclic organic carbonates under scalable continuous flow conditions”
Z. Wang, R. Gérardy, G. Gauron, C. Damblon and J.-C. M. Monbaliu*
React. Chem. Eng. 2019, 4, 17-26
“Sustaining the transition from petro- to biobased chemical industry with flow chemistry”
R. Gérardy, R. Morodo, J. Estager, P. Luis, D. P. Debecker, and J.-C. M. Monbaliu*
Top. Curr. Chem. 2019, 377:1
Publications of 2018
“A versatile biobased continuous flow strategy for the production of 3-butene-1,2-diol and vinyl ethylene carbonate from erythritol”
N. Ntumba Tshibalonza, R.Gérardy, Z. Alsafra, G. Eppe and J.-C. M. Monbaliu*
Green Chem. 2018, 20, 5147-5157
Part of the themed collection: 2018 Green Chemistry Hot Articles
“Improving Continuous Flow Singlet Oxygen Photooxygenations with Functionalized Mesoporous Silica Nanoparticles”
C. Mendoza, N. Emmanuel, C. A. Páez, L. Dreesen*, J.-C. M. Monbaliu* and B. Heinrich*
ChemPhotoChem 2018, 2, 890-897
Part of the special issue: Flow photochemistry
“Application of pervaporation in the bio-production of glycerol carbonate”
W. Li, R. Sreerangappa, J. Estager, J.-C. M. Monbaliu, D. P. Debecker, P. Luis
Chem. Eng. Process. 2018, 132, 127-136
“Sorption and pervaporation study of methanol/diméthyl carbonate mixture with poly(etheretherketone) (PEEK-WC) membrane”
W. Li,F. Galiano, J. Estager, J.-C. M. Monbaliu, D. P. Debecker, A. Figoli, P. Luis
J. Memb. Sci. 2018, 567, 303-310
“Accelerated microfluidic native chemical ligation at difficult amino acids toward cyclic peptides”
N. Ollivier, T. Toupy, R. C. Hartkoorn, R. Desmet, J.-C. M. Monbaliu* and O. Melnyk*
Nat. Commun. 2018, 9, 2847
DOI: 10.1038/s41467-018-05264-8
Highlighted in: SYNFACTS of the month
Featured in: 2018 Synthetic and Medicinal Chemistry issue of Nature Communication
“Continuous Flow Organic Chemistry: Successes and Pitfalls at the Interface with Current Societal Challenges”
R. Gérardy, N. Emmanuel, T. Toupy, V. Kassin, N. Ntumba Tshibalonza, M. Schmitz, J.-C. M. Monbaliu*
Eur. J. Org. Chem. 2018, 2301-2351
Tagged as Very Important Paper
Most Accessed paper in June 2018
Featured in the special Collection EurJOC Readers’ Choice 2019
“NOTA–PRGD2 and NODAGA–PRGD2: Bioconjugation, Characterization, Radiolabelling & Design Space”
M. Salvé, H. T. Avohou, J.-C. M. Monbaliu, P. Lebrun, C. Lemaire, C. Damblon, P. de Tullio, R. Hustinx and A. Luxen
J. Label. Cmpd. Radiopharm. 2018, 61, 487-500
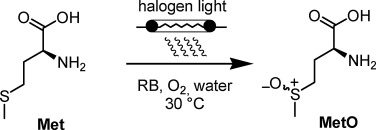
“Transitioning from macroscopic batch to microfluidic processes for the efficient singlet oxygen photooxygenation of methionine”
C. Mendoza, N. Emmanuel, C. Paez, L. Dreesen*, J.-C. M. Monbaliu* and B. Heinrichs*
J. Photochem. Photobiol. 2018, 356,193-200
Publications of 2017
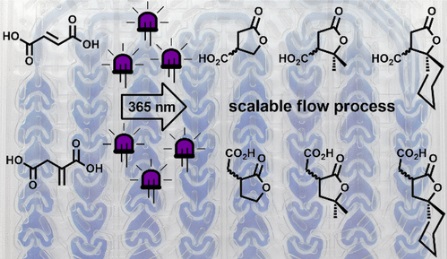
“Continuous-flow preparation of g-butyrolactone scaffolds from renewable fumaric and itaconic acids under photosensitized conditions”
R. Gérardy, M. Winter, C. R. Horn., A. Vizza, K. Van Hecke and J.-C. M. Monbaliu*
Org. Process Res. Dev. 2017, 21, 2012–2017
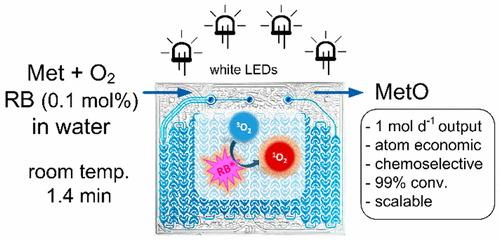
“Scalable Photocatalytic Oxidation of Methionine under Continuous-Flow Conditions”
N. Emmanuel, C. Mendoza, M. Winter, C. Horn, A. Vizza, L. Dreesen, B. Heinrichs and J.-C. M. Monbaliu*
Org. Process Res. Dev. 2017, 21, 1435-1438
“Revisiting the deoxydehydration of glycerol towards allyl alcohol under continuous-flow conditions”
N. Ntumba Tshibalonza and J.-C. M. Monbaliu*
Green Chem. 2017, 19, 3006-3013
“Exploring the Fundamentals of Microreactor Technology with Multidisciplinary Lab Experiments Combining the Synthesis and the Characterization of Inorganic Nanoparticles”
N. Emmanuel, G. Emonds-Alt, M. Lismont*, G. Eppe* and J.-C. M. Monbaliu*
J. Chem. Edu. 2017, 94, 775–780
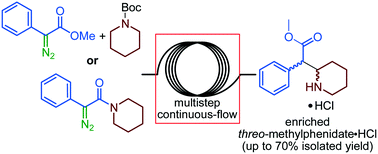
“Assessing inter- and intramolecular continuous-flow strategies towards methylphenidate (Ritalin) hydrochloride”
R. Gérardy, M. Winter, A. Vizza and J.-C. M. Monbaliu*
React. Chem. Eng. 2017, 2, 149-158
Featured in: the themed collection Reaction Chemistry & Engineering Emerging Investigators
2016
- “Expanding chemistry’s horizon with continuous-flow reactors” J.-C. M. Monbaliu*, N. Emmanuel and R. Gérardy, Chimie Nouvelle 2016, 122, 18-26 (here)
- “Compact and integrated approach for advanced end-to-end production, purification, and aqueous formulation of lidocaine hydrochloride”, J.-C. M. Monbaliu, T. Stelzer, E. Revalor, N. Weeranoppanant, K. F. Jensen, A. S. Myerson, Org. Process Res. Dev. 2016, 20, 1347-1353 (doi: 10.1021/acs.oprd.6b00165)
- “On-demand continuous flow production of pharmaceuticals in a compact, reconfigurable system”, A. Adamo, R. L. Beingessner, M. Behnam, J. Chen, T. F. Jamison, K. F. Jensen, J.-C. M. Monbaliu, A. S. Myerson, E. Revalor, D. R. Snead, T. Stelzer, N. Weeranoppanant, S. Y. Wong, P. Zhang, Science 2016, 352, 61-67 (doi: 10.1126/science.aaf1337)
Highlighted on STAT, Technobahn, Science News, IEEE, MIT News, Scientific American, HNGN, MedicalXPress, Technology Review, Chemical & Engineering News
- “Continuous-flow N-heterocyclic carbenes generation and organocatalysis”, L. Di Marco, M. Hans, L. Delaude and J.-C. M. Monbaliu*, Chem. Eur. J. 2016, 22, 4508–4514 (doi: 10.1002/chem.201505135)
Tagged as hot paper and highlighted on Chemistry Views (here)
- “Accelerating chemoselective peptide bond formation using bis(2-selenylethyl)amido peptide selenoester surrogates”, L. Raibaut, M. Cargoët, N. Ollivier, Y.-M. Chang, H. Drobecq, E. Boll, R. Desmet, J.-C. M. Monbaliu* and Oleg Melnyk*, Chem. Sci. 2016, 7, 2657-2665 (doi: 10.1039/C5SC03459K)
2015
- “Continuous-flow thermolysis for the preparation of vinylglycine derivatives”, N. Lamborelle, J. Simon, A. Luxen and J.-C. M. Monbaliu*, Org. Biomol. Chem. 2015, 13, 11602-11606 (doi: 10.1039/C5OB02036K)
- “Preparation, reactivity and synthetic utility of simple benzotriazole derivatives”, R. Gérardy and J.-C. M. Monbaliu*, Top. Heterocycl. Chem. 2015, 1-66 (doi: 110.1007/7081_2015_179)
- “Efficient continuous flow benzotriazole activation and coupling of amino acids”, S. Seghers, F. E. A. Van Waes, A. Cukalovic, J.-C. M. Monbaliu et al., J. Flow Chem., 2015, 5, 220-227 (doi: 10.1556/1846.2015.00029)
2014
- “New benzotriazole-based reagents for the phosphonylation of various N-, O- and S-nucleophiles”, Pandmand, A. D. Tiwari, S. Panda, J.-C. M. Monbaliu et al., Tetrahedron Lett. 2014, 55, 5898-5901 (doi: 10.1016/j.tetlet.2014.07.057)
- “L’intensification des procédés chimiques, une approche radicale”, C. Henneuse, J.-C. M. Monbaliu, Athena 2014, 18-21 (here)
- “Feruloyl benzotriazole and Weinreb amide as bioinspired building blocks: a reactivity study towards O-, N-, S- and C-nucleophiles”, B. I. Roman, J.-C. M. Monbaliu et al., Eur. J. Org. Chem. 2014, 2594-2611. (doi: 10.1002/ejoc.201301895)
2013
- “Capture of benzotriazole-based Mannich electrophiles by CH-acidic compounds”, J.-C. M. Monbaliu, L. K. Beagle et al., RSC Advances 2013, 3, 4152-4155 (doi: 10.1039/C3RA22826F)
- “Governing parameters of long-range intramolecular S-to-N acyl transfers within chemical ligation of peptides”, J.-C. M. Monbaliu, G. Dive et al., J. Chem. Theory Comp. 2013, 927-934 (doi: 10.1021/ct300830k)
- “Development, optimization and scale-up of biodiesel production from crude palm oil and effective use in developing countries”, A. Cukalovic, J.-C. M. Monbaliu et al., Biomass & Bioenerg. 2013, 56, 62-69 (doi: 10.1016/j.biombioe.2013.04.015)
2012
- “Recent trends in Cys- and Ser/Thr-based chemical ligations for the elaboration of peptide constructs”, J.-C. M. Monbaliu and A. R. Katritzky, Chem. Commun. 2012, 48, 11601-11622 (doi: 10.1039/C2CC34434C)
- “A convenient synthesis of difficult medium-sized cyclic peptides by Staudinger-mediated ring closure”, K. Ha, J.-C. M. Monbaliu et al., Org. Biomol. Chem. 2012, 10, 8055-8058 (doi: 10.1039/C2OB25996F)
- “En route towards ?-benzotriazoyl nitroso derivatives”, J.-C. M. Monbaliu, Lucas Beagle et al., RSC Advances 2012, 2, 8941-8945 (doi: 10.1039/C2RA21311G)
- “Efficient synthesis of 2,5-diketopiperazines by Staudinger-mediated cyclization”, L. Beagle, F. Hansen, J.-C. M. Monbaliu et al., Synlett 2012, 23, 2337-2340 (doi: 10.1055/s-0031-1290446)
- “User-friendly and flexible Kiliani-Fisher reaction on ketoses using microreaction technology”, J.-C. M. Monbaliu, A. Cukalovic et al., J. Flow Chem. 2012, 2, 43-46 (doi: 10.1556/JFC-D-12-00003)
- “Long-range intramolecular S-N acyl migration: a study of the formation of native peptide analogs via 13-, 15- and 16-membered cyclic transition states”, K. Ha, M. Chahar, J.-C. M. Monbaliu et al., J. Org. Chem. 2012, 77, 2637-2648 (doi: 10.1021/jo2023125)
- “A new benzotriazole-mediated stereoflexible gateway to hetero-2,5-diketopiperazines”, J.-C. M. Monbaliu, F. K. Hansen et al., Chem. Eur. J. 2012, 18, 2632-2638 (doi: 10.1002/chem.201103143)
2011
- “Continuous-flow production of alkyl nitrites”, J.-C. M. Monbaliu*, J. Jorda et al., Chemistry Today 2011, 29, 80-82 (here)
- “Electron-deficient 1- and 2-aza-1,3-dienes: a comprehensive survey of their synthesis and reactivity
J.-C. M. Monbaliu, K. G. R. Masschelein and C. V. Stevens, Chem. Soc. Rev. 2011, 40, 4708-4739 (doi: 10.1039/C1CS15070G)
- “Effective production of the biodiesel additive STBE by a continuous flow process”, J.-C. M. Monbaliu, M. Winter et al., Bioresour. Technol. 2011, 102, 9304-9307 (doi: 10.1016/j.biortech.2011.07.007)
2010
- “Straightforward hetero Diels-Alder cycloadditions of nitroso dienophiles by microreactor technology”,J.-C. M. Monbaliu, A. Cukalovic et al., Tetrahedron Lett. 2010, 51, 5830-5833 (doi: 10.1016/j.tetlet.2010.08.117)
- “Feasibility study for industrial production of fuel additives from glycerol”,
J.-C. M. Monbaliu, M. Winter et al., Chemistry Today 2010, 28, 42-45 (here)
- “[4+2] Cycloaddition of 1-phosphono-1,3-butadienes with nitroso-heterodienophiles: a new and versatile entry to polyfunctionalized ?– and ?-aminophosphonic derivatives”, J.-C. M. Monbaliu, B. Tinant et al, J. Org. Chem. 2010, 75, 5478-5486 (doi: 10.1021/jo100230r)
- “HD-A cycloadditions of 1-diethoxyphosphoryl-1,3-butadiene with nitroso- and azo-heterodienophiles: a theoretical investigation”, J.-C. M. Monbaliu*, G. Dive et al., J. Mol. Struct.: THEOCHEM. 2010, 959, 49-54 (doi: 10.1016/j.theochem.2010.08.004)
- “Three unrelated sphingomyelin analogs spontaneously cluster into plasma membrane micrometric domains”, D. Tyteca, L. D’Auria, P. Van Der Smissen, T. Medts, S. Carpentier, J.-C. M. Monbaliu et al., BBA-Biomembrane 2010, 1978, 909-927 (doi: 10.1016/j.bbamem.2010.01.021)
- “Novel chiral 1-phosphono-1,3-butadiene for asymmetric hetero Diels-Alder cycloadditions with nitroso and azodicarboxylate dienophiles”, J.-C. M. Monbaliu, D. Peeters et al., Tetrahedron Lett. 2010, 51, 1052-1055 (doi: 10.1016/j.tetlet.2009.12.063)
2009
- “Is anthracene cofactor or spectator for the thermolysis of anthracenyl acylnitroso cycloadducts in the presence of a diene?”, J.-C. M. Monbaliu*, J. Marchand-Brynaert et al., Tetrahedron Lett. 2009, 50, 2555-2558 (doi: 10.1016/j.tetlet.2009.03.062)
- “A practical synthesis of 3-diethoxyphosphoryl-1,2-pyridazine derivatives”, J.-C. M. Monbaliu, J. Marchand-Brynaert, Synthesis 2009, 11, 1876-1880 (doi: 10.1055/s-0028-1088056)
- (R)-4-phenyloxazolidin-2-thione: an efficient chiral auxiliary for [4+2] cycloaddition of 1-aminodienes and activated phosphonodienophiles, J.-C. M. Monbaliu, R. Robiette et al, Tetrahedron Lett. 2009, 50, 1314-1317 (doi: 10.1016/j.tetlet.2009.01.036)
2008
- “Reactivity of (R)-4-phenyloxazolidin-2-thione chiral auxiliary: from deprotection to heterocyclic interconversion”, J.-C. M. Monbaliu, B. Tinant et al., Heterocycles 2008, 75, 2459-2475 (doi: 10.3987/COM-08-11410)
- “[4+2] Cycloaddition of 1-phosphono-1,3-butadiene with azo- and nitroso-heterodienophiles”, J.-C. M. Monbaliu and J. Marchand-Brynaert, Tetrahedron Lett. 2008, 49, 1839-1842 (doi: 10.1016/j.tetlet.2008.01.050)
- “NMR and X-ray diffraction analysis of 3-thioamido-5-phosphono-1-cyclohexene derivatives: Conformational and stereochemical assignments”, J.-C. M. Monbaliu, B. Tinant et al, J. Mol. Struct. 2008, 879, 113-118 (doi: 10.1016/j.molstruc.2007.08.018)



![[2026][447x216] images_large_an5c05506_0007 [2026][447x216] images_large_an5c05506_0007](http://www.citos.uliege.be/wp-content/uploads/2026/01/2026447x216-images_large_an5c05506_0007-300x145.jpg)
![[2025][447x232]TOC_law [2025][447x232]TOC_law](http://www.citos.uliege.be/wp-content/uploads/2025/12/2025447x232TOC_law-300x156.jpg)
![[2025][447x303]TOC JACS Au [2025][447x303]TOC JACS Au](http://www.citos.uliege.be/wp-content/uploads/2025/12/2025447x303TOC-JACS-Au-300x203.jpg)
![[2025][447x450]TOC [2025][447x450]TOC](http://www.citos.uliege.be/wp-content/uploads/2025/12/2025447x450TOC-298x300.jpg)
![[2025][447x446]TOC ChemSusChem [2025][447x446]TOC ChemSusChem](http://www.citos.uliege.be/wp-content/uploads/2025/12/2025447x446TOC-ChemSusChem-300x300.jpg)
![[2025][447x173]1-s2.0-S0378517325010348-ga1_lrg [2025][447x173]1-s2.0-S0378517325010348-ga1_lrg](http://www.citos.uliege.be/wp-content/uploads/2025/12/2025447x1731-s2.0-S0378517325010348-ga1_lrg-300x116.jpg)
![[2025][447x241]image(36) [2025][447x241]image(36)](http://www.citos.uliege.be/wp-content/uploads/2025/12/2025447x241image36-300x162.jpg)
![[2025][447x97] Angew. Chem. Int. Ed. 2025, e202501660 [2025][447x97] Angew. Chem. Int. Ed. 2025, e202501660](http://www.citos.uliege.be/wp-content/uploads/2025/05/2025447x97-Angew.-Chem.-Int.-Ed.-2025-e202501660-300x65.jpg)
![[2025][447x203]React. Chem Eng. 2025 [2025][447x203]React. Chem Eng. 2025](http://www.citos.uliege.be/wp-content/uploads/2025/05/2025447x203React.-Chem-Eng.-2025-300x136.jpg)
![[2025][447x422]ChemPhotoChem 2025 [2025][447x422]ChemPhotoChem 2025](http://www.citos.uliege.be/wp-content/uploads/2025/05/2025447x422ChemPhotoChem-2025-300x283.jpg)
![[2025][447x181]1-s2.0-S0927796X25000178-ga1_lrg [2025][447x181]1-s2.0-S0927796X25000178-ga1_lrg](http://www.citos.uliege.be/wp-content/uploads/2025/05/2025447x1811-s2.0-S0927796X25000178-ga1_lrg-300x121.jpg)
![[2025][447x163] chem202403701-toc-0001-m [2025][447x163] chem202403701-toc-0001-m](http://www.citos.uliege.be/wp-content/uploads/2025/05/2025447x163-chem202403701-toc-0001-m-300x109.jpg)
![[2025][447x266]Int. J. Pharm. 2025, 670, 125070 [2025][447x266]Int. J. Pharm. 2025, 670, 125070](http://www.citos.uliege.be/wp-content/uploads/2025/05/2025447x266Int.-J.-Pharm.-2025-670-125070-300x179.jpg)
![[2025][447x226] Rapid CO2 [2025][447x226] Rapid CO2](http://www.citos.uliege.be/wp-content/uploads/2025/01/2025447x226-Rapid-CO2-300x152.jpg)
![[2024][447x226] ToC_final(2) [2024][447x226] ToC_final(2)](http://www.citos.uliege.be/wp-content/uploads/2024/08/2024447x226-ToC_final2-300x152.png)
![[2024] image(31) [2024] image(31)](http://www.citos.uliege.be/wp-content/uploads/2024/08/2024-image31-300x199.png)
![[2024] image(30) [2024] image(30)](http://www.citos.uliege.be/wp-content/uploads/2024/08/2024-image30-300x123.png)
![[2024][447x241] [2024][447x241]](http://www.citos.uliege.be/wp-content/uploads/2024/08/2024447x241-300x162.png)
![[2024][447x224] Abstract_3 [2024][447x224] Abstract_3](http://www.citos.uliege.be/wp-content/uploads/2024/08/2024447x224-Abstract_3-300x150.png)
![[2024][447x241] image(29) [2024][447x241] image(29)](http://www.citos.uliege.be/wp-content/uploads/2024/08/2024447x241-image29-300x162.png)
![[2024][447x331] RSC_cover_AT(1) [2024][447x331] RSC_cover_AT(1)](http://www.citos.uliege.be/wp-content/uploads/2024/08/2024447x331-RSC_cover_AT1-300x222.png)
![[2024][447x210] TOC_glycerol carbonate [2024][447x210] TOC_glycerol carbonate](http://www.citos.uliege.be/wp-content/uploads/2024/01/2024447x210-TOC_glycerol-carbonate-300x141.jpg)
![[2024][447x192] image(26) [2024][447x192] image(26)](http://www.citos.uliege.be/wp-content/uploads/2024/01/2024447x192-image26-300x129.jpg)
![[2023][443x209] Warfarin [2023][443x209] Warfarin](http://www.citos.uliege.be/wp-content/uploads/2023/11/2023443x209-Warfarin-300x142.jpg)
![[2022][447x447] A perspective on automated advanced continuous flow manufacturing [2022][447x447] A perspective on automated advanced continuous flow manufacturing](http://www.citos.uliege.be/wp-content/uploads/2022/12/2022447x447-A-perspective-on-automated-advanced-continuous-flow-manufacturing-300x300.jpg)
![[2022][447x86] Continuous Flow Photooxidative Degradation of Azo [2022][447x86] Continuous Flow Photooxidative Degradation of Azo](http://www.citos.uliege.be/wp-content/uploads/2022/12/2022447x86-Continuous-Flow-Photooxidative-Degradation-of-Azo-300x58.jpg)
![[447x341]On Demand Flow Platform [447x341]On Demand Flow Platform](http://www.citos.uliege.be/wp-content/uploads/2022/10/447x341On-Demand-Flow-Platform.jpg)
![[2022][447x252] Accelerating the End [2022][447x252] Accelerating the End](http://www.citos.uliege.be/wp-content/uploads/2022/09/2022447x252-Accelerating-the-End.jpg)
![[2026][447x175]1-s2.0-S0039914022004362-ga1_lrg [2026][447x175]1-s2.0-S0039914022004362-ga1_lrg](http://www.citos.uliege.be/wp-content/uploads/2026/03/2026447x1751-s2.0-S0039914022004362-ga1_lrg.jpg)
![[2022][447x219] A Continuous Flow Generator [2022][447x219] A Continuous Flow Generator](http://www.citos.uliege.be/wp-content/uploads/2022/04/2022447x219-A-Continuous-Flow-Generator.jpg)
![[2022][447x87] JFlowChem [2022][447x87] JFlowChem](http://www.citos.uliege.be/wp-content/uploads/2022/04/2022447x87-JFlowChem.jpg)
![[2022][447x216] Modafinil [2022][447x216] Modafinil](http://www.citos.uliege.be/wp-content/uploads/2026/03/2022447x216-Modafinil.jpg)
![[2022][447x136] Intensified Continuous Flow [2022][447x136] Intensified Continuous Flow](http://www.citos.uliege.be/wp-content/uploads/2026/03/2022447x136-Intensified-Continuous-Flow.jpg)

![[378x51] [378x51]](http://www.citos.uliege.be/wp-content/uploads/2021/12/378x51.jpg)
![[2021][447x224]graphical_abstract-nitroso-pauline [2021][447x224]graphical_abstract-nitroso-pauline](http://www.citos.uliege.be/wp-content/uploads/2021/12/2021447x224graphical_abstract-nitroso-pauline.jpg)
![[2021][447x176] Propellant, Explosive [2021][447x176] Propellant, Explosive](http://www.citos.uliege.be/wp-content/uploads/2021/10/2021447x176-Propellant-Explosive.jpg)
![[2021][447x359] Flow neutralization of sulfur-containing [2021][447x359] Flow neutralization of sulfur-containing](http://www.citos.uliege.be/wp-content/uploads/2021/04/2021447x359-Flow-neutralization-of-sulfur-containing.jpg)
![[2021][447x238]A Modular_2 [2021][447x238]A Modular_2](http://www.citos.uliege.be/wp-content/uploads/2021/02/2021447x238A-Modular_2.png)
![[2020][447x225]] Au Nanobipyr [2020][447x225]] Au Nanobipyr](http://www.citos.uliege.be/wp-content/uploads/2021/02/2020447x225-Au-Nanobipyr-1.png)
![[2020][447x447] Continuous Flow Upgrading [2020][447x447] Continuous Flow Upgrading](http://www.citos.uliege.be/wp-content/uploads/2020/07/2020447x447-Continuous-Flow-Upgrading-300x300.png)
![[2020][447x451] Continuous Flow Organophosphorus [2020][447x451] Continuous Flow Organophosphorus](http://www.citos.uliege.be/wp-content/uploads/2020/07/2020447x451-Continuous-Flow-Organophosphorus-297x300.png)



![[2020][447x339] Separation of bio_based [2020][447x339] Separation of bio_based](http://www.citos.uliege.be/wp-content/uploads/2020/05/2020447x339-Separation-of-bio_based-1.jpg)
![[2020] Radical C-H F-Difluoromethylation of [447x285] [2020] Radical C-H F-Difluoromethylation of [447x285]](http://www.citos.uliege.be/wp-content/uploads/2020/03/2020-Radical-C-H-F-Difluoromethylation-of-447x285.jpg)
![[2019] Metal-free hydroxylation [447x168] [2019] Metal-free hydroxylation [447x168]](http://www.citos.uliege.be/wp-content/uploads/2019/12/2019-Metal-free-hydroxylation-447x168.jpg)
![[2020] Supported ionic liquid membranes for the separation [447x310] [2020] Supported ionic liquid membranes for the separation [447x310]](http://www.citos.uliege.be/wp-content/uploads/2020/02/2020-Supported-ionic-liquid-membranes-for-the-separation-447x310.jpg)
![[2019] Allentoine3 [447x216] [2019] Allentoine3 [447x216]](http://www.citos.uliege.be/wp-content/uploads/2019/12/2019-Allentoine3-447x216.jpg)
![[2019] Versatile and scalable synthesis of cyclic organic carbonates [447x193] [2019] Versatile and scalable synthesis of cyclic organic carbonates [447x193]](http://www.citos.uliege.be/wp-content/uploads/2019/10/2019-Versatile-and-scalable-synthesis-of-cyclic-organic-carbonates-447x193.png)
![[2019] Understanding chemical interaction [447x223] [2019] Understanding chemical interaction [447x223]](http://www.citos.uliege.be/wp-content/uploads/2019/08/2019-Understanding-chemical-interaction-447x223.jpg)
![[2019] Continuous flow upgrading of glycerol toward [447x122] [2019] Continuous flow upgrading of glycerol toward [447x122]](http://www.citos.uliege.be/wp-content/uploads/2019/07/2019-Continuous-flow-upgrading-of-glycerol-toward-447x122.jpg)
![[2019] Expedient Preparation of Active Pharmaceutical Ingredient Ketamine under Sustainable Continuous Flow Conditions [2019] Expedient Preparation of Active Pharmaceutical Ingredient Ketamine under Sustainable Continuous Flow Conditions](http://www.citos.uliege.be/wp-content/uploads/2019/03/2019-Ket.jpg)
![[2019] Native chemical ligation and extended methodologies [2019] Native chemical ligation and extended methodologies](http://www.citos.uliege.be/wp-content/uploads/2019/05/2019-Native-chemical-ligation-and-extended-methodologies-300x293.jpg)
![[2019] Solubility Determination and Correlation of Warfarin [447x234] [2019] Solubility Determination and Correlation of Warfarin [447x234]](http://www.citos.uliege.be/wp-content/uploads/2019/05/2019-Solubility-Determination-and-Correlation-of-Warfarin-447x234.jpeg)
![[2019] Finding the perfect match [447x150] [2019] Finding the perfect match [447x150]](http://www.citos.uliege.be/wp-content/uploads/2019/01/2019-Finding-the-perfect-match-447x150.jpg)
![[2018] Solvent-free organocatalytic [447x269] [2018] Solvent-free organocatalytic [447x269]](http://www.citos.uliege.be/wp-content/uploads/2018/10/2018-Solvent-free-organocatalytic-447x269.png)
![[2018] sustaining the transition[447x269] [2018] sustaining the transition[447x269]](http://www.citos.uliege.be/wp-content/uploads/2018/10/2018-sustaining-the-transition447x269.jpg)
![[2018] Production of 3-butene-1,2-diol and vinyl ethylene carbonate from erythritol [447x269] [2018] Production of 3-butene-1,2-diol and vinyl ethylene carbonate from erythritol [447x269]](http://www.citos.uliege.be/wp-content/uploads/2018/10/2018-Production-of-3-butene-12-diol-and-vinyl-ethylene-carbonate-from-erythritol-447x269.jpg)
![[2018] Improving Continuous Flow Singlet [447x269] [2018] Improving Continuous Flow Singlet [447x269]](http://www.citos.uliege.be/wp-content/uploads/2018/10/2018-Improving-Continuous-Flow-Singlet-447x269.jpg)
![[2018] Application of pervaporation [447x269] [2018] Application of pervaporation [447x269]](http://www.citos.uliege.be/wp-content/uploads/2018/10/2018-Application-of-pervaporation-447x269.jpg)
![[2018] Sorption and pervaporation study [447x269] [2018] Sorption and pervaporation study [447x269]](http://www.citos.uliege.be/wp-content/uploads/2018/10/2018-Sorption-and-pervaporation-study-447x269.jpg)
![[2018] Accelerated microfluidic [447x269] [2018] Accelerated microfluidic [447x269]](http://www.citos.uliege.be/wp-content/uploads/2018/08/2018-Accelerated-microfluidic-447x269.jpg)
![[V1] Cover Feature [500x500] [V1] Cover Feature [500x500]](http://www.citos.uliege.be/wp-content/uploads/2018/08/V1-Cover-Feature-500x500-300x300.jpg)